Process Behavior Charts: Separating Behavior from Specification
Right now there is a long running debate on whether specification limits should be shown on Process Behavior Charts (control charts).
Meeting specification keeps you compliant today. Understanding variation keeps you compliant tomorrow.
Those following Shewhart and Wheeler make a crucial distinction: process behavior charts reveal how a process performs over time, while specification limits reflect external requirements—they are not part of the chart and should not be used to interpret it.
A Process Behavior Chart answers one question. What is the process doing over time. It separates routine variation from signals that require action.
Specifications answer a different question: is the result acceptable? In pharmaceuticals, specifications define quality requirements agreed with regulators and reflect the safety and efficacy expected for the patient. They define the outcome at the point of assessment, where the samples used are intended to be representative of the entire batch, not how the process behaves over time, including the variation used to establish control limits.
Control limits serve a different role. They are derived from the process and define action limits. They signal when investigation or intervention adds economic value. When the process remains within these limits, the correct action is to do nothing. The most common failure with decision makers is not recognizing this distinction.
Control limits improve performance by enabling correct decisions. They prevent unnecessary adjustment, focus attention on real signals, and trigger improvement when needed. This allows the process to reach and sustain its full design potential.
Problems begin when both specifications and control limits are shown without a defined purpose. Given that specifications are often wider than control limits, decision makers tend to default to specifications. They overlook the economic value of respecting control limits and rely on acceptance outcomes alone. The focus shifts to acceptance rather than process behaviour, and decisions are driven by proximity to specification rather than signals in the data.
The question is not whether specifications should be shown.
The question is what you are trying to learn.
If you want to understand the process, specifications distract
If you want to communicate risk or conformance, specifications add a time sensitive context
The objective defines whether process behavior chart should have specification limits present.
What Process Behavior Charts are used for?
Control charts are used to:
Monitor and control process behavior
Understand variation and enable prediction
Support investigation and root cause analysis
Verify improvement
Communication - Report state of control to work colleagues, customers, suppliers, regulators with OPV/CPV and APR/PQR
Compare variation against specification over time
Define rational subgrouping and sampling strategies
The objective determines the Process Behavior Charts design
For operators, charts must be simple and actionable. Use:
Target
Upper and lower control limits
Mechanism to capture Actions taken
These reflect real process variation and define when action is justified.
Do not include specifications. They do not help the operator run the process, as replacing control limits only increases the frequency of false signals and unnecessary actions.
For reporting or review, include both when needed:
Control limits to show behavior
Specification limits to show acceptability, but make sure the audience understands the difference otherwise they will default to the widest limits shown.
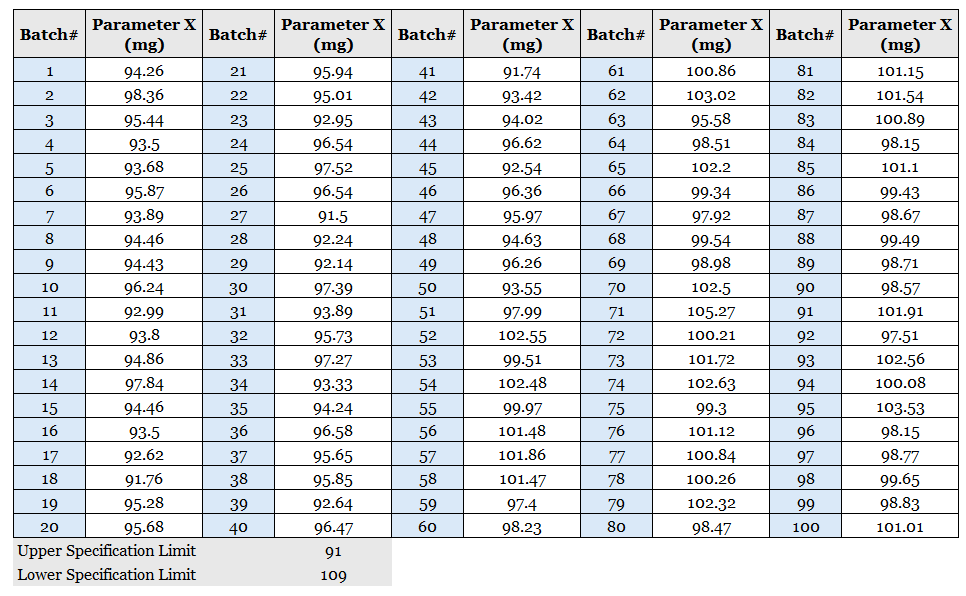
Table 1: Historical values of Parameter X (mg)
Table 1, data often sits in rows and columns. The only check applied is whether values fall within specification.
In this example, all batches pass.
From a compliance perspective, this is often presented as individuals argue that the specifications are consistently met.
It is argued that a state of control is demonstrated.
Under ICH Q10 Pharmaceutical Quality System, a state of control requires that the process consistently provides assurance of performance and product quality
That should mean:
Stable behavior
Predictable output
Evidence over time
A table cannot show this. It hides sequence, variation, and change.
All results can pass while the process remains unknown.
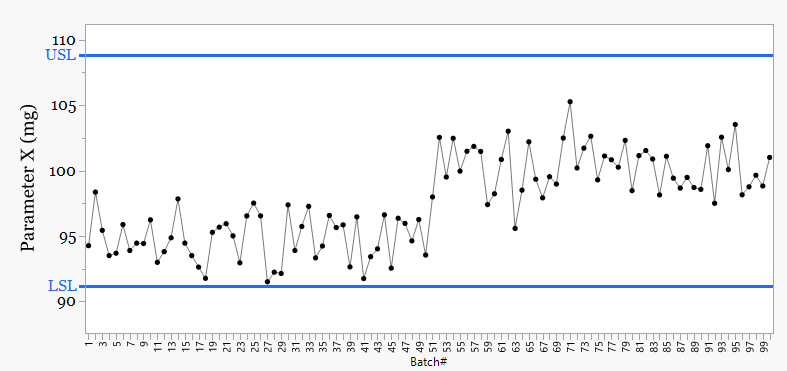
Figure 1: Specifications only
Figure 1 now plots the data and a pattern appears.
A shift is visible.
Without control limits, the change can be seen but not evaluated. There is no rule for action, only interpretation.
With more subtle step changes or less obvious non-random patterns, rule-based decision making becomes necessary.
It is only when we move to Figure 2 and apply a rule-based method that judgement is replaced.
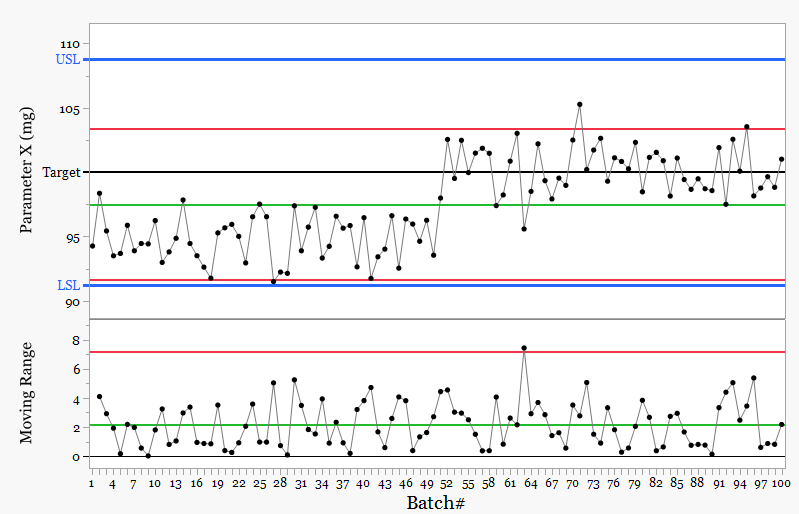
Figure 2. Both specification and control limits
With Figure 2, adding control limits changes the picture. The process shifts, with more than seven consecutive points above the mean. Rule based decisions identify this as a signal. The focus moves from individual results to process behavior, and action is directed at the underlying cause rather than isolated values.
This type of chart is often used for reporting, not for assessing fast moving manufacture. It often leads to a familiar but incorrect conclusion:
Everything is in specification
Therefore the process is in control
Here, “in control” is interpreted as within specification, not statistical control. The process is compliant, but not stable.
This interpretation misses the point.
Reality is:
The chart shows two different process states.
The mean blends them. It represents neither - it does not represent anything
Cpk around 1.08 suggests acceptable capability. That calculation assumes a single stable process. That assumption is false.
You are averaging two behaviors and reporting one number.
The message is misleading.
The process and its variation meets specification.
A StepChange is present, yet everything remains within specification.
No action is taken
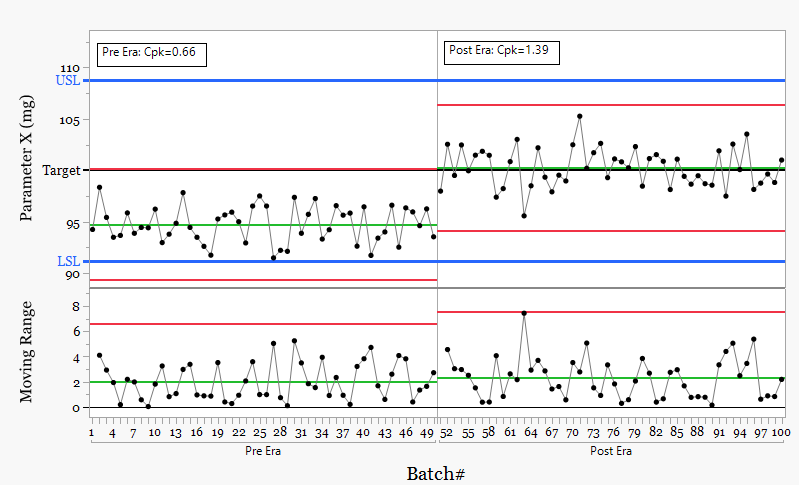
Figure 3. Subgrouping by process state
Split the data into Pre and Post.
Now the process is clear.
Two states. Two behaviors. Two different risks.
Conclusions
1. Control limits and Cpk across the full dataset are misleading. They describe an average that often does not exist in practice.
2. In the Pre Era, control limits fall below the LSL. The process produces values below specification even though none were observed. Cpk of 0.66 confirms the risk.
3. The process is off target. Meeting specification hides the loss associated with this position.
4. The step change is real. It must be explained. This is process knowledge, not an inconvenience.
5. In the Post Era, the process is centered and stable. Control limits sit within specification. Cpk improves to around 1.39.
6. Specification limits support communication. Control limits support control. Use them accordingly.
Perspective
At Verto we assess a process by the position of the control limits relative to specification.
Cpk and Ppk can mislead when the process is not stable or when used without the chart. The timeline shows what the process is doing. We will use these indices but only when they reflect the process relationship with the specifications.
When designing process behavior charts for manufacturing control, we avoid including specifications. They add complexity without supporting decision making.
Rational subgrouping is critical. If you group data correctly, the truth appears.
Specifications on histograms support analysis but are insensitive to time and that matters.
Summary
– when to include specifications on process behavior charts
Know the objective
Know your audience
Decide whether specifications support or distract from the objective and the audience
Is there value in showing how the relationship between process variation and specifications changes over time?